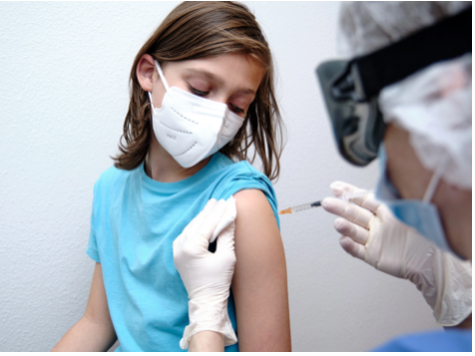
Pfizer Plans to Release COVID-19 Vaccine for Kids
October 6, 2021
Pfizer and BioNtech hope to release the long awaited COVID vaccine for kids ages 5-11 very soon after trials and promising results.
On Tuesday October 28, Pfizer submitted their clinical trial to the Food and Drug Administration for the new Covid vaccine targeted for children ages 5 to 11.
The initial test from the trial which studied the effectiveness and personal safety of the Pfizer COVID-19 vaccine for children ages 5 to 11 showed promising results.
Pharmaceutical companies stated that early results of the Pfizer trial prove the vaccine is safe for children and sets a strong response against the virus. COVID-19 vaccines for young children get closer every day. As their arrival is highly anticipated, the vaccine is undeniably a potential game-changer for all families, schools, and communities.
“Considering I have an 11-year-old brother, I think at the beginning people will be very nervous about letting their kids and siblings get the vaccine. Although maybe after a month or so when more people start to get it, they will feel it’s safe. I would let my brother get the vaccine because I feel the best scientists are working on it and their trials have shown good results,” said sophomore Hannah Brewster.
The trial used a smaller dosage of 10 micrograms, instead of the 30 microgram dose used for people 12 and older. The reports also state side effects were generally stable compared to older people who already received the vaccine. It is said shots will officially be available for kids approximately in mid November.
“Hopefully, this means we are one step closer to ending this pandemic and finally being able to go back to normal. My 8-year-old cousin has been having difficulty being online, quarantining and actually paying attention so I am glad we can finally move past this horrid time and make progress,” said sophomore Mariandrea Robelo.
Presently, Moderna and Johnson & Johnson are still testing their shots for young children in the U.S. Roughly 28 million Americans fall between the ages of 5 and 11. Allowing them to finally be eligible for Covid vaccines will permit children to attend school in person more safely. It’ll be a positive development for the rest of the country, as well as it’ll raise national vaccination rates and lower parental stress levels.